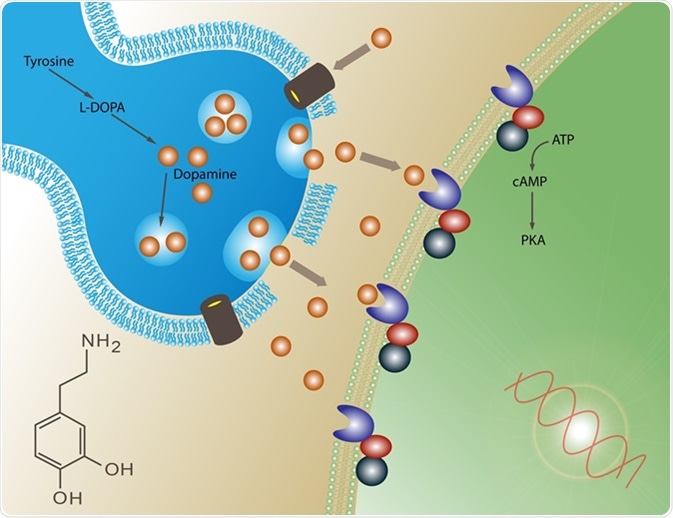
Unlocking GPCR Signaling: The Dynamic Dance of G-Protein Recognition and Activation
G-protein-coupled receptors (GPCRs) are crucial cellular signaling hubs, but how they selectively recognize and activate different G-proteins has remained a mystery. A groundbreaking 2026 Nature study using time-resolved cryo-electron microscopy reveals the dynamic structural basis of this process. By capturing over 20 intermediate states of the neurotensin receptor 1 (NTSR1) interacting with Gi, Go, and Gq proteins, researchers have visualized the stepwise molecular choreography of G-protein association, activation, and dissociation. This research provides unprecedented insights into GPCR signaling dynamics, offering new pathways for developing more precise therapeutic drugs targeting these essential cellular receptors.
G-protein-coupled receptors (GPCRs) represent one of the largest and most important families of membrane proteins in the human body, responsible for translating extracellular signals into cellular responses. Despite decades of research, the dynamic molecular mechanisms governing how GPCRs recognize and activate different G-protein subtypes have remained elusive. A landmark 2026 study published in Nature, titled "The dynamic basis of G-protein recognition and activation by a GPCR," has fundamentally advanced our understanding of these processes through unprecedented time-resolved structural visualization.

The Structural Revolution in GPCR Research
Traditional structural biology has provided over 200 static snapshots of GPCR-G protein complexes, but these frozen moments offered limited insight into the dynamic processes of association and dissociation. The recent breakthrough, as detailed in the Nature publication, employed time-resolved cryo-electron microscopy to capture the molecular dance between the neurotensin receptor 1 (NTSR1) and various G-protein subtypes. This approach allowed researchers to visualize intermediate states that were previously invisible, revealing how the receptor's intracellular surface dynamically rearranges to accommodate different G-proteins.
Key Mechanistic Discoveries
Dynamic Receptor Rearrangement
The study demonstrated that NTSR1 employs common intracellular rearrangements to recognize different G-protein subtypes while simultaneously promoting activation of a single subtype. This dual functionality represents a sophisticated molecular adaptation that enables precise cellular signaling. The receptor's flexibility allows it to interact with multiple G-protein partners while maintaining specificity in downstream signaling pathways.
Distinct Dissociation Pathways
One of the most significant findings was that Gi dissociates from the receptor through a pathway distinct from that of Gs. Furthermore, the canonical and non-canonical NTSR1–Gi complexes diverge in their dissociation trajectories. This discovery challenges previous assumptions about uniform G-protein dissociation mechanisms and suggests that different receptor-G protein pairs may employ unique separation strategies.

Stepwise G-Protein Activation
The research identified four key mechanistic features through characterization of more than 20 intermediates, complemented by mutational and computational analyses. First, GDP/GTP induces Gi release from both canonical and non-canonical active conformations with distinct kinetics. Second, separation from Gβγ involves stepwise remodeling of the Gα switches I–III. These switches act as molecular relays that transmit conformational changes throughout the G-protein structure.
Nucleotide-Induced Conformational Changes
The study revealed how GDP and GTP binding trigger specific conformational changes in the G-protein. When GDP binds, it stabilizes the inactive state, while GTP binding promotes the active conformation that dissociates from the receptor. The time-resolved analysis captured these transitions in unprecedented detail, showing how the G-protein's α-helical domain (AHD) opens and closes during activation.
Implications for Therapeutic Development
These findings provide a comprehensive framework for understanding GPCR signaling dynamics that could revolutionize drug development. By understanding the precise molecular steps of G-protein recognition and activation, researchers can design more selective therapeutic compounds. The study's insights into how receptors accommodate different G-protein subtypes while activating specific pathways offer new opportunities for developing drugs with fewer side effects.
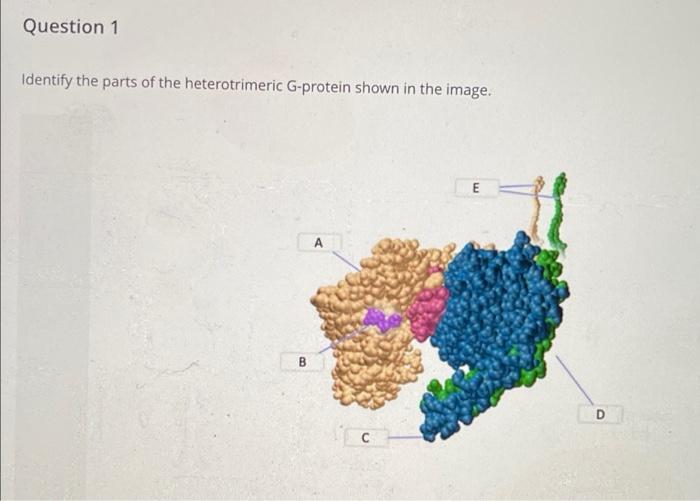
Methodological Advancements
The research employed cutting-edge cryo-electron microscopy techniques, including 3D variability analysis (3DVA) to capture stepwise GDP-induced G-protein dissociation from the receptor. The team analyzed structures of human neurotensin receptor type 1 (NTSR1) with minimally modified Go and Gq proteins, revealing how the receptor's intracellular surface dynamically rearranges to accommodate each G-protein subtype. All cryo-EM density maps and atomic coordinates have been deposited in public databases, making this valuable structural information available to the scientific community.
Future Directions in GPCR Research
This research opens new avenues for investigating other GPCR-G protein pairs and their signaling dynamics. The methodological framework established by this study can be applied to understand how different receptors achieve signaling specificity and how disease mutations might disrupt these delicate molecular interactions. As noted in the publication, these findings "provide a comprehensive framework for understanding GPCR signalling dynamics and guiding signal-targeted therapeutic development."
The dynamic visualization of GPCR-G protein interactions represents a paradigm shift in structural biology, moving from static snapshots to molecular movies that capture the fluid nature of cellular signaling. As researchers continue to apply these advanced techniques to other receptor systems, our understanding of cellular communication will deepen, potentially leading to breakthroughs in treating numerous diseases that involve GPCR signaling pathways.