How Pancreatic Cancer Reprograms Its Environment to Spread Stealthily
Pancreatic cancer's lethality is partly due to its ability to manipulate surrounding healthy tissue to facilitate early and aggressive spread. A groundbreaking study from Brazilian researchers reveals the central role of a protein called periostin in this process. By remodeling the extracellular matrix and aiding cancer cell invasion into nerves, periostin creates pathways for metastasis and builds a protective barrier that shields tumors from treatment. This discovery opens new avenues for targeted therapies aimed at blocking this invasion mechanism before it begins, offering hope for improving the grim prognosis associated with this disease.
Pancreatic cancer remains one of the most formidable challenges in oncology, notorious for its aggressive nature and poor survival rates. Its lethality is not merely a product of rapid tumor growth but stems from a sophisticated ability to hijack and reprogram the body's own tissues to enable stealthy, early spread. A pivotal study published in Molecular and Cellular Endocrinology has illuminated a key mechanism behind this deadly progression, focusing on a protein called periostin. This research, conducted by scientists in Brazil, provides a clearer understanding of how pancreatic tumors remodel their microenvironment, invade critical structures like nerves, and ultimately evade treatment, while also pointing to a promising new therapeutic target.
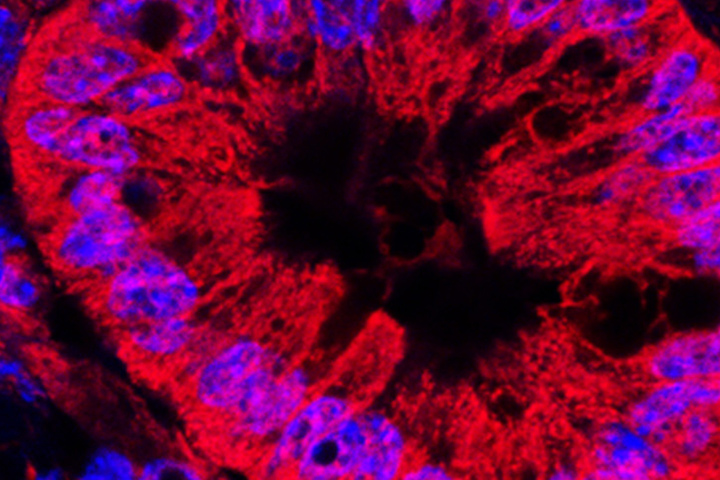
The Deadly Dynamics of Pancreatic Cancer
Pancreatic adenocarcinoma, which accounts for roughly 90% of pancreatic cancer cases, develops in the glandular cells responsible for producing digestive enzymes. Globally, it is diagnosed in about 510,000 people each year, with a nearly equivalent number of annual deaths, underscoring its aggressive trajectory. In Brazil, estimates indicate approximately 11,000 new cases and 13,000 deaths yearly. The prognosis is stark, with only around 10% of patients surviving five years post-diagnosis. This poor outlook is intrinsically linked to the cancer's capacity for early and widespread metastasis, a process fueled by its manipulation of the local tissue environment.
Unveiling the Role of Periostin in Tissue Remodeling
The study's central finding involves the protein periostin and its production by pancreatic stellate cells. Periostin is known for its role in reshaping the extracellular matrix—the structural network that organizes and supports healthy tissue. In pancreatic cancer, tumor cells co-opt this function. The research demonstrates that periostin participates in extensive tissue remodeling, effectively "paving the way" for cancer cells to break through their local confines. This disruption involves specialized enzymes and creates a path of least resistance, allowing malignant cells to reach and infiltrate nearby anatomical structures.
Nerve Invasion: A Highway for Metastasis
A critical consequence of this tissue remodeling is perineural invasion, a hallmark of pancreatic cancer aggressiveness. This process occurs when cancer cells enter and spread along nerve bundles. Nerves act as natural conduits connecting different body regions, providing tumor cells with a direct route to travel far from the primary site and establish metastases. More than half of pancreatic cancer cases show evidence of this nerve invasion at an early stage. Unfortunately, it is typically only identified after surgery during biopsy analysis, by which point the invasive process is already advanced. This early spread significantly contributes to the disease's high mortality rate.
The Treatment-Resistant Barrier
As the tumor reprograms its surroundings, it triggers a desmoplastic reaction. This involves the accumulation of dense, fibrous, and inflamed tissue around the cancer cells, forming a stiff, protective barrier. This hardened microenvironment does more than just facilitate invasion; it actively impedes treatment. The fibrous stroma makes it exceedingly difficult for chemotherapy drugs and immunotherapies to penetrate and reach the tumor cells in effective concentrations. This protective shield allows cancer cells to survive, proliferate, and continue their invasive spread, rendering many conventional therapies less effective.
Targeting Periostin: A Path to New Therapies
The identification of periostin's role offers a tangible target for future therapeutic strategies. The researchers suggest that inhibiting periostin activity or depleting the stellate cells that produce it could disrupt the early stages of tissue remodeling and nerve invasion. This approach aligns with the broader shift toward precision medicine in oncology. By preventing the tumor from acquiring its invasive capacity so early, treatments could potentially contain the disease and improve outcomes. Clinical trials investigating antibodies designed to block periostin are already underway for other cancers, and their findings may inform similar approaches for pancreatic cancer.

Conclusion and Future Directions
The discovery of how pancreatic cancer uses periostin to remodel its environment and invade nerves represents a significant advance in understanding this deadly disease. It moves beyond viewing the tumor in isolation to recognizing the active, supportive role played by the reprogrammed tissue surrounding it. This insight not only explains the cancer's aggressive behavior and treatment resistance but also opens a new frontier for intervention. Future research and drug development focused on blocking this invasion pathway before it starts hold the promise of transforming the therapeutic landscape for pancreatic cancer, potentially turning it from a swift and stealthy killer into a more manageable condition.




