How Pancreatic Cancer Hijacks Tissue to Spread: The Periostin Pathway
Pancreatic cancer's lethality stems from its ability to spread early and stealthily. A groundbreaking 2026 study reveals how the tumor reprograms its microenvironment using a protein called periostin. This process facilitates nerve invasion, creates a protective fibrous barrier that resists treatment, and paves the way for metastasis. Understanding this mechanism opens new avenues for targeted therapies aimed at blocking invasion before it starts, offering hope for improving the dire prognosis associated with this aggressive disease.
Pancreatic cancer remains one of the most formidable challenges in oncology, notorious for its aggressive spread and poor survival rates. A pivotal 2026 study published in Molecular and Cellular Endocrinology has illuminated a critical mechanism behind this lethality. Researchers discovered that pancreatic tumors actively reprogram their surrounding tissue, using a specific protein to invade nerves and create a fortress-like barrier. This revelation not only explains why the disease is so difficult to treat but also points to a promising new target for intervention.
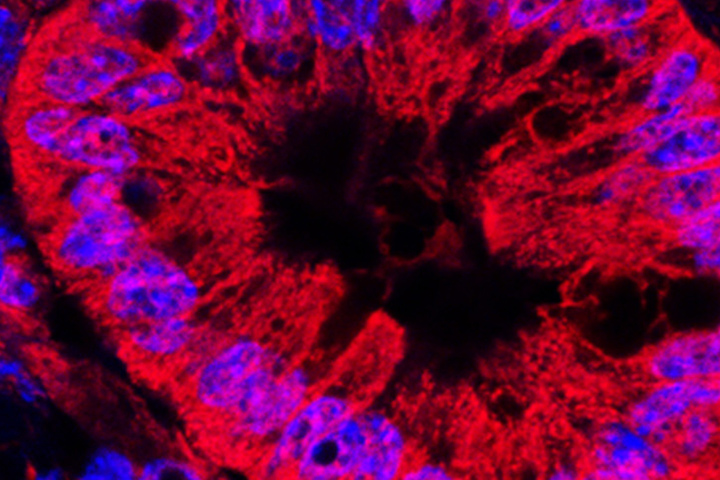
The Deadly Dynamics of Pancreatic Adenocarcinoma
The most common form, pancreatic adenocarcinoma, originates in the glandular cells. It accounts for approximately 90% of cases and is characterized by a death rate that nearly matches its diagnosis rate. Globally, this translates to about 510,000 new cases and a similar number of deaths annually. In Brazil, where the research was conducted, estimates indicate 11,000 new cases and 13,000 deaths each year. "It's an aggressive cancer that's difficult to treat. Around 10% of patients have a chance of long-term survival," explains study author and oncologist Dr. Pedro Luiz Serrano Uson Junior, highlighting the urgent need for better therapeutic strategies.
Unraveling the Mechanism of Early Spread
The study's core finding centers on a process known as perineural invasion. This occurs when cancer cells infiltrate and travel along nerves, a hallmark of aggressiveness that facilitates metastasis and often causes severe pain. "Perineural invasion is a marker of cancer aggressiveness," Uson states. The research team, led by Carlos Alberto de Carvalho Fraga and principal investigator Helder Nakaya at the Center for Research on Inflammatory Diseases (CRID), used advanced spatial genomics to analyze 24 tumor samples. They found the cancer does not act alone; it co-opts the stroma—the connective tissue surrounding the tumor—turning it from a passive structure into an active accomplice in invasion.
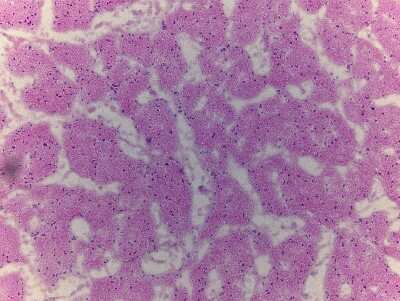
The Key Player: Periostin and Tissue Remodeling
The orchestrator of this hostile takeover is a protein called periostin, produced abundantly by pancreatic stellate cells. Periostin's primary function is to remodel the extracellular matrix—the scaffold that holds tissues together. In the context of cancer, this remodeling becomes destructive. "Periostin participates in this remodeling, paving the way for tumor cells to invade," explains Nakaya. By breaking down and reorganizing the tissue architecture, periostin essentially clears a path for cancer cells to reach nearby nerves, which then act as highways for further dissemination throughout the body.
The Dual Challenge: Invasion and Treatment Resistance
This invasive process triggers a desmoplastic reaction, resulting in the buildup of dense, fibrous tissue around the tumor. This hardened barrier serves a dual purpose: it protects the cancer cells and severely impedes the delivery of chemotherapy and immunotherapy drugs. "That's why pancreatic cancer is still so difficult to treat," Uson notes. The study indicates that more than half of pancreatic cancer cases show signs of this nerve invasion at an early stage, but it is typically only discovered post-surgery, after the spread has already occurred.
A New Frontier in Precision Oncology
The identification of periostin's role presents a tangible new target for therapy. The researchers propose that inhibiting periostin or depleting the stellate cells that produce it could prevent early nerve invasion and slow metastasis. This approach aligns with the shift toward precision medicine. "If we can develop antibodies or drugs that block these stellate cells, we'll have tools to prevent the tumor from acquiring this invasive capacity so early," says Uson. Notably, clinical trials for other cancers are already testing anti-periostin antibodies, which could pave the way for pancreatic cancer applications.
This research, supported by FAPESP, underscores a critical advance. By understanding how pancreatic cancer rewires its microenvironment, the medical community moves closer to developing pre-emptive strategies. The goal is to intercept the disease before it spreads, ultimately transforming the prognosis for patients facing this daunting diagnosis.