Brain-Immune Crosstalk: The Hidden Culprit Worsening Heart Attack Damage
Recent research reveals a surprising connection between the brain and the immune system that exacerbates damage following a heart attack. Studies in mice show that specific neurons in the vagus nerve create a damaging feedback loop between the heart and brain, triggering harmful inflammatory responses. This discovery opens new pathways for therapeutic interventions that could significantly improve recovery outcomes for heart attack patients by targeting this neural-immune axis.
When we think of heart attacks, we typically imagine blocked arteries and damaged heart muscle. However, groundbreaking research published in Cell reveals a more complex picture involving intricate communication between the heart, brain, and immune system. This discovery of a heart-brain-immune axis suggests that the body's own protective mechanisms might actually worsen cardiac damage following a myocardial infarction.
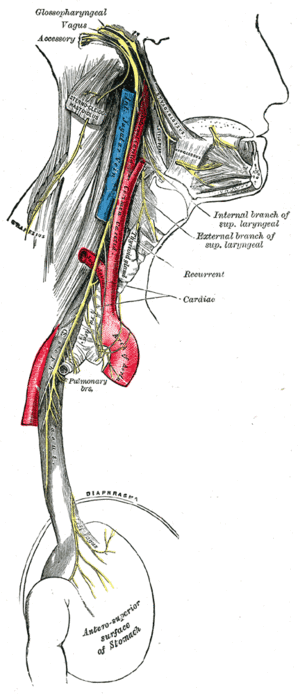
The Heart-Brain-Immune Axis Explained
During a heart attack, researchers discovered that a specific set of neurons within the vagus nerve—the body's primary communication highway between organs and the brain—becomes hyperactive. These neurons relay distress signals from the damaged heart to the brain, which in turn triggers widespread immune and inflammatory responses. This frenzied crosstalk creates a vicious cycle where the initial damage is amplified by the body's own defense mechanisms.
The vagus nerve, traditionally known for its role in regulating heart rate and digestion, appears to have a previously unrecognized function in coordinating immune responses to cardiac injury. When these neural pathways are activated during a myocardial infarction, they initiate inflammatory processes that extend far beyond the initial site of damage, potentially affecting healthy heart tissue and complicating recovery.
Potential Therapeutic Implications
Perhaps the most promising aspect of this research comes from the experimental interventions. When researchers blocked these specific neural signals in mouse models, they observed significantly improved outcomes following heart attacks. This suggests that targeted therapies could potentially interrupt this damaging feedback loop, offering a new approach to heart attack treatment beyond traditional methods focused solely on restoring blood flow.
The discovery opens several potential therapeutic avenues. Pharmaceutical approaches might target the specific neurotransmitters or receptors involved in this neural-immune communication. Neuromodulation techniques, such as vagus nerve stimulation with specific parameters, could potentially be refined to selectively inhibit the damaging signals while preserving beneficial neural functions. These approaches represent a paradigm shift in how we conceptualize heart attack treatment—moving beyond immediate mechanical interventions to address the body's systemic response to cardiac injury.
Broader Scientific Context
This research emerges alongside other significant scientific developments that highlight the interconnected nature of biological systems. The study of disruptive camouflage in blue manakins, where birds decorate nests with elaborate tails to confuse predators, demonstrates how organisms evolve sophisticated defense mechanisms. Similarly, the heart-brain-immune axis represents an evolved response that, in the context of modern cardiac events, may have become maladaptive.
The findings also intersect with growing concerns about research integrity in the digital age. As highlighted in recent analyses, social media platforms like X (formerly Twitter) have become early warning systems for problematic scientific papers, with critical posts often preceding formal retractions. This underscores the importance of robust scientific discourse and verification, particularly for groundbreaking discoveries like the heart-brain-immune connection that could lead to new therapeutic approaches.

Future Research Directions
While the mouse study provides compelling evidence, significant questions remain before this research can translate to human therapies. Researchers must determine whether the same neural-immune mechanisms operate in humans, what specific molecular pathways are involved, and whether blocking these signals has any unintended consequences for other bodily functions. Additionally, timing will be crucial—therapeutic interventions would need to be administered within specific windows following a heart attack to be effective.
Future studies will likely explore how this neural-immune axis interacts with other known factors in heart attack recovery, including genetic predispositions, comorbidities like diabetes or hypertension, and lifestyle factors. Understanding these interactions could lead to personalized approaches to heart attack treatment that account for individual variations in neural and immune responses.
Conclusion
The discovery of the heart-brain-immune axis represents a significant advancement in our understanding of cardiac pathology. By revealing how neural communication can exacerbate heart attack damage, this research opens new possibilities for therapeutic intervention that could complement existing treatments. As we continue to unravel the complex interactions between our nervous, cardiovascular, and immune systems, we move closer to more comprehensive approaches to heart health that address not just the mechanical aspects of cardiac events, but the body's holistic response to injury.