The Molecular Switch: How Alzheimer's May Trick Neurons into Erasing Memory
New research from Stanford University suggests Alzheimer's disease may cause memory loss by hijacking a single molecular receptor that tells neurons to prune their own synaptic connections. This groundbreaking study reveals that both amyloid beta proteins and inflammatory molecules converge on the same pathway, triggering excessive synapse elimination. The findings challenge traditional views of Alzheimer's progression and point toward new therapeutic strategies that could protect memory by targeting this specific receptor mechanism.
Alzheimer's disease represents one of the most devastating neurological challenges of our time, steadily dismantling the intricate neural networks that form our memories and cognitive abilities. For decades, researchers have pursued various theories about how this destruction begins, with amyloid beta plaques and inflammation emerging as leading suspects. Now, a groundbreaking study from Stanford University's Wu Tsai Neurosciences Institute reveals these two factors may converge on a single molecular switch that actively tells neurons to erase their own connections.

The Discovery of a Shared Pathway
The research, published in Proceedings of the National Academy of Sciences, centers on a receptor known as LilrB2. This molecule has been studied for years by lead researcher Carla Shatz, who previously discovered its role in normal synaptic pruning during brain development. In 2013, her team made a crucial connection: amyloid beta binds to LilrB2, triggering neurons to remove synapses. What's remarkable about the new findings is that inflammatory molecules appear to use the same pathway.
Scientists screened complement cascade molecules—part of the immune system's inflammatory response—and found that the protein fragment C4d binds strongly to LilrB2. When researchers injected C4d into the brains of healthy mice, the results were startling. "Lo and behold, it stripped synapses off neurons," Shatz reported, describing a surprising effect for a molecule previously thought to have no function. This suggests that both amyloid beta and inflammation may drive synapse loss through the same biological mechanism, potentially explaining why Alzheimer's progresses through multiple pathways.

Neurons as Active Participants
One of the most significant implications of this research challenges a fundamental assumption in Alzheimer's science. For years, many researchers believed that glial cells—the brain's immune cells—were primarily responsible for removing synapses in the disease. This study suggests neurons themselves play a more direct and active role in their own destruction.
"Neurons aren't innocent bystanders," Shatz emphasized. "They are active participants." This paradigm shift suggests that Alzheimer's may involve neurons responding to signals that normally help shape brain connections during development and learning, but which become destructive when activated excessively or inappropriately in the aging brain.
Therapeutic Implications and Future Directions
The discovery has profound implications for Alzheimer's treatment strategies. Currently, FDA-approved medications focus primarily on breaking apart amyloid plaques, an approach that has yielded limited benefits and significant side effects, including headaches and brain bleeding. "Busting up amyloid plaques hasn't worked that well," Shatz noted, adding that even successful amyloid removal addresses only part of the problem.
Targeting the LilrB2 receptor offers a potentially more comprehensive approach. By protecting synapses directly—the actual sites where memories are stored and processed—researchers hope to preserve memory function itself. This strategy could complement existing approaches or provide an alternative for patients who don't respond to current therapies. The research received support from the Knight Initiative for Brain Resilience, a program specifically focused on reexamining the basic biology behind neurodegenerative diseases.
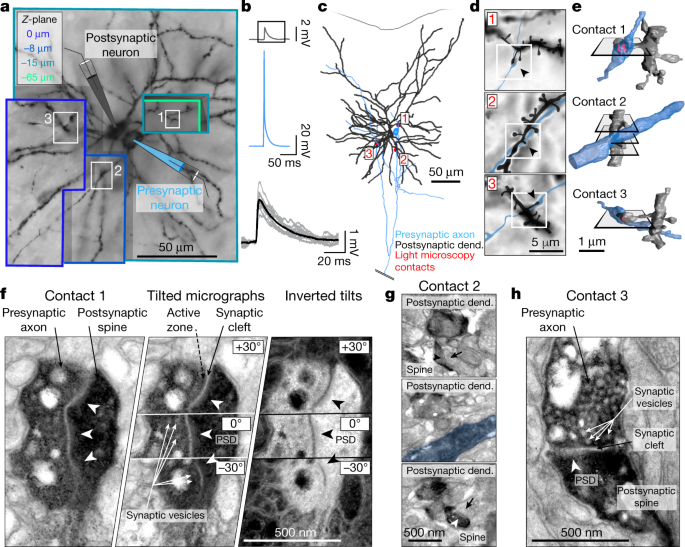
Connecting Major Alzheimer's Theories
Perhaps the most exciting aspect of this research is how it connects two major theories of Alzheimer's development. The amyloid hypothesis has dominated research for decades, while inflammation has gained increasing attention as a contributing factor. The discovery that both converge on LilrB2 provides a unifying framework that could explain how multiple risk factors lead to the same devastating outcome: memory loss.
"There's an entire set of molecules and pathways that lead from inflammation to synapse loss that may not have received the attention they deserve," said Shatz, who serves as both a professor of biology and neurobiology at Stanford. This integrated understanding could accelerate drug development by identifying points in the pathway where interventions might be most effective.
Conclusion: A New Direction for Alzheimer's Research
The Stanford research represents a significant step forward in understanding Alzheimer's fundamental mechanisms. By identifying LilrB2 as a convergence point for multiple pathological processes, scientists now have a clearer target for developing therapies that protect synapses and preserve memory. While much work remains to translate these findings into clinical treatments, the study offers hope for more effective approaches that address the disease's complexity.
As research continues, focusing on how neurons actively participate in their own decline may unlock new strategies for intervention. The study underscores the importance of looking beyond amyloid plaques alone and considering the broader biological context of Alzheimer's disease. With continued investigation into receptors like LilrB2 and their role in synaptic maintenance, we move closer to treatments that could meaningfully alter the course of this devastating condition.