Lesion-Remote Astrocytes: Orchestrators of White Matter Repair After Spinal Cord Injury
Groundbreaking research published in Nature reveals how lesion-remote astrocytes (LRAs) in the spinal cord orchestrate critical white matter repair processes after injury. These specialized astrocytes, located in spared regions away from the primary injury site, undergo distinct molecular transformations and secrete the protein CCN1 to direct microglia-mediated clearance of myelin debris. This cellular coordination is essential for neurological recovery, with astrocyte CCN1 signaling regulating microglial lipid metabolism and debris processing. The study demonstrates how this astrocyte-microglia axis represents a fundamental, evolutionarily conserved repair mechanism that could be therapeutically targeted across multiple central nervous system disorders.
Spinal cord injury represents one of the most challenging neurological conditions, with limited treatment options and often permanent functional deficits. While much research has focused on the immediate injury site, a groundbreaking study published in Nature reveals that cells far from the primary lesion play a surprisingly critical role in recovery. Lesion-remote astrocytes (LRAs), specialized star-shaped glial cells in spared spinal cord regions, undergo remarkable molecular transformations that orchestrate white matter repair through sophisticated communication with microglia, the brain's immune cells.

The Distinct Nature of Lesion-Remote Astrocytes
Following spinal cord injury, astrocytes in regions adjacent to the lesion form a protective barrier, but researchers have discovered that astrocytes in more distant, spared regions undergo entirely different transformations. Using sophisticated transcriptional profiling methods, scientists investigated LRAs from spared regions of mouse spinal cord following traumatic injury. They found that LRAs acquire a spectrum of molecularly distinct, neuroanatomically restricted reactivity states that evolve over time after injury.
Unlike border-forming astrocytes that create physical barriers, LRAs intermingle with viable neurons and glia throughout cytoarchitecturally intact regions. These cells exhibit varying degrees of cellular hypertrophy but undergo little to no proliferation. The molecular and functional properties of these LRAs remained largely undefined until this research revealed their crucial role in directing tissue repair processes.
White Matter Astrocytes and the CCN1 Signaling Pathway
The study identified a particularly important subset of white matter LRAs that express the secreted matricellular protein CCN1. These CCN1-expressing astrocytes are specifically induced by local myelin damage and are found throughout degenerating white matter regions. Remarkably, these astrocytes persist for at least 90 days post-injury, indicating their sustained role in the repair process.
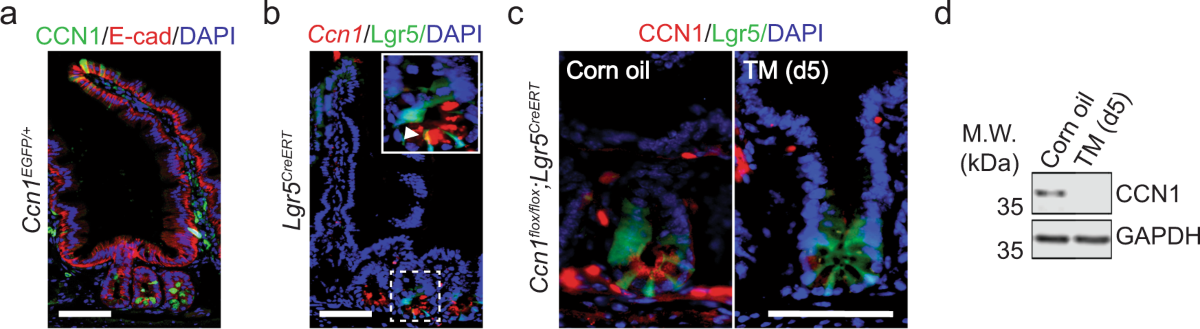
Astrocyte-derived CCN1 serves as a critical signaling molecule that governs the specification and function of local microglia. When researchers created mice with conditional astrocyte-specific CCN1 deletion (Ccn1-cKO mice), they observed dramatic consequences for white matter repair. Loss of astrocyte CCN1 resulted in excessive, aberrant activation of local microglia, characterized by abnormal molecular specification and impaired debris processing.
Microglial Dysfunction Without Astrocyte Guidance
In the absence of astrocyte CCN1 signaling, microglia exhibited intracellular accumulation of myelin and axon debris alongside dysregulated lipid metabolism with distinctive attenuation in lipid droplet accumulation. This dysfunction culminated in blunted clearance of white matter debris and impaired neurological recovery from spinal cord injury. The research demonstrates that CCN1 binds to microglial SDC4 receptors to augment lipid storage, linking this signaling axis to a vital repair-associated lipid buffering response in debris-clearing microglia.
The Astrocyte-Microglia Repair Axis
The study reveals a sophisticated cellular communication system where white matter LRAs direct the molecular, metabolic, and functional specification of debris-clearing microglia. This astrocyte-microglia axis represents a fundamental repair mechanism that supports neurological recovery after injury. The researchers used computational modeling of LRA-mediated heterotypic cell interactions, astrocyte-specific gene deletion, and multiple mouse models of acute and chronic CNS white matter degeneration to unravel this previously unrecognized repair pathway.
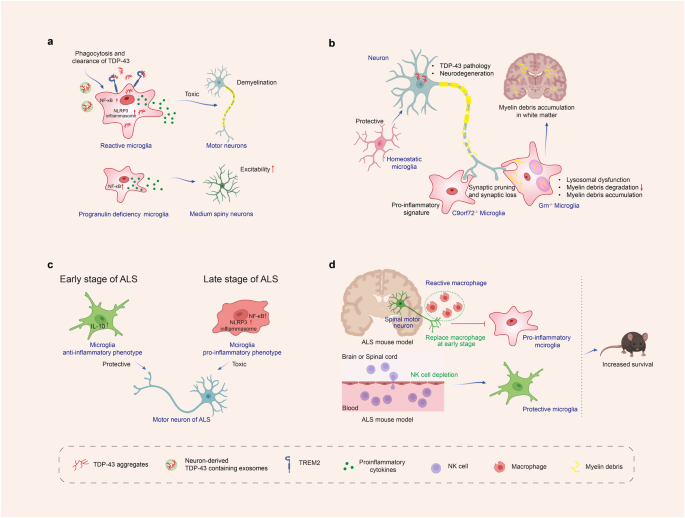
White matter degeneration-associated microglia (WDM) gradually assemble into multicellular nodules that phagocytose lipid-rich myelin debris to promote tissue repair. These WDM intermingle intimately with CCN1-expressing LRAs, forming a functional unit for debris clearance. The spatial relationship between these cell types is so precise that WDM nodules are significantly more likely to be proximate to CCN1+ astrocytes than would be expected by random chance.
Clinical Implications and Evolutionary Conservation
The significance of this discovery extends beyond spinal cord injury. CCN1-expressing white matter astrocytes are generated in diverse demyelinating disorders in both mice and humans, pointing to their fundamental, evolutionarily conserved role in white matter repair. The researchers found CCN1+ astrocytes in mouse models of lysolecithin-mediated demyelination and experimental autoimmune encephalomyelitis (EAE), as well as in human spinal cord tissue from individuals with multiple sclerosis or spinal cord injury.
In human tissue, CCN1-expressing astrocytes were frequent in white matter associated with multiple sclerosis lesions and lesion-remote normal-appearing white matter, but rare in multiple sclerosis-associated grey matter and healthy spinal cord. This conservation across species and conditions suggests that targeting this astrocyte-microglia axis could have broad therapeutic applications for various white matter disorders.
Mechanistic Insights and Therapeutic Potential
The research provides detailed mechanistic understanding of how astrocyte CCN1 regulates microglial function. CCN1 directly modulates microglial lipid metabolism gene expression and promotes an adaptive lipid buffering response characterized by lipid droplet accumulation and reduced cholesterol efflux capacity. This metabolic reprogramming supports efficient uptake and intracellular digestion of lipid-rich debris.

When microglia were stimulated with CCN1 in culture, they showed prominent upregulation of TREM2-dependent gene signatures previously linked to white matter repair, debris clearance, and neuroprotection. CCN1 treatment also enhanced lipid uptake and expression of lipid storage genes while suppressing genes required for lipid efflux. These findings position LRAs as key orchestrators of multicellular neural repair processes that promote functional recovery after CNS injury.
Future Directions and Clinical Translation
The discovery of this astrocyte-microglia repair axis opens new avenues for therapeutic development. The researchers suggest that manipulating LRA reactivity states may be a viable path for limiting chronic neuroinflammation, enhancing functionally meaningful regenerative plasticity, and promoting neurological recovery after CNS injury and in disease. Since CCN1 expression represents an evolutionarily conserved astrocyte response to myelin degeneration, this pathway could be therapeutically harnessed across a spectrum of CNS disorders.
The study's findings strongly support further investigation into whether astrocyte CCN1-mediated signaling can be enhanced to improve clearance of white matter debris, restrict inflammation, and promote white matter repair. As researchers continue to unravel the complex cellular interactions in spared regions of the injured CNS, these insights will shape next-generation treatments that manipulate these dynamic cellular landscapes to promote neural repair and functional recovery.