How Childhood Immune Imprinting Shapes Future Flu Protection
New research reveals how a child's first influenza infections can permanently shape their immune system's ability to fight future flu strains. The study, published in Nature, demonstrates that B cell imprinting in children specifically impairs antibody responses to the conserved haemagglutinin stalk—a key target for universal flu vaccines. This phenomenon, known as original antigenic sin, causes the immune system to preferentially recall its initial response to related pathogens, potentially reducing protection against evolving strains. Understanding these mechanisms is crucial for developing more effective vaccination strategies, particularly for children whose immune systems are forming lifelong patterns of response.
When a child encounters their first influenza virus, their immune system makes a lasting impression—one that could shape their ability to fight flu for decades to come. A groundbreaking study published in Nature reveals how B cell imprinting in young children specifically impairs antibody responses to the haemagglutinin stalk, a conserved region of the influenza virus that represents a prime target for universal vaccine development. This phenomenon, first described as "original antigenic sin" and now understood as immune imprinting, has profound implications for how we approach influenza vaccination and protection across the lifespan.

Understanding Immune Imprinting
Immune imprinting, also known as original antigenic sin, refers to the immune system's tendency to preferentially recall its initial response to a pathogen when encountering related strains later in life. This phenomenon has been observed with various viruses but is particularly significant for influenza due to the virus's rapid evolution and antigenic drift. The Nature study provides crucial insights into how this process unfolds at the cellular level, specifically examining B cell responses in children experiencing their first influenza infections.
The research team investigated young children aged 2–6 years who experienced sequential first-time exposures to heterosubtypic influenza viruses—specifically H3N2 followed by H1N1 infections, or vice versa. By comparing these responses to those of adults and children with single infections, researchers uncovered fundamental differences in how immune memory forms during these critical early exposures.

The Haemagglutinin Stalk Dilemma
Current influenza vaccines primarily stimulate antibodies against the globular head of the haemagglutinin protein, which mediates viral attachment to host cells. However, this region is highly mutagenic, allowing influenza viruses to rapidly evolve and escape immune detection. In contrast, the haemagglutinin stalk—which mediates membrane fusion—is highly conserved across influenza strains but traditionally less immunogenic.
The stalk region has emerged as a promising target for universal influenza vaccines because antibodies against this conserved area could potentially provide broad protection against multiple strains. However, the new research reveals a significant challenge: childhood imprinting specifically impairs the development of effective antibodies against this crucial region.
Mechanisms of Impaired Antibody Responses
The study employed sophisticated techniques including LIBRA-seq (linking B cell receptor to antigen specificity by sequencing) and single-cell RNA sequencing to track how B cells respond to sequential influenza infections. Researchers found that after consecutive heterosubtypic primary infections, 4–6% of memory B cells in children become cross-reactive between H1 and H3 strains, targeting the conserved central stalk epitope.
However, these imprinted B cells exhibit a critical flaw: over 90% show higher affinity for the first infecting strain (the imprinting strain) compared to subsequent strains. This affinity bias results in reduced breadth and neutralization potency against later influenza viruses. The researchers identified a single amino acid change in the stalk epitope (D46N) as central to this nearly universal shift in reactivity—despite differing by only a single atomic group between carboxylic acid and amide.
Childhood Versus Adult Immune Responses
The research reveals fundamental differences between how children and adults develop influenza immunity. While children's primary B cell responses are phenotypically similar to adults', with subtle gene expression differences, adult B cells show substantial evidence of homosubtypic imprinting—preferential responses to strains within the same influenza subtype encountered earlier in life.
Adult B cells commonly cross-react with past strains using more stereotyped and mutated immunoglobulin genes, indicating extensive refinement through repeated exposures. In contrast, children's responses after sequential heterosubtypic infections show minimal memory recall and robust de novo responses, but with the concerning imprinting effect on stalk-targeting antibodies.
Structural Insights into Imprinting
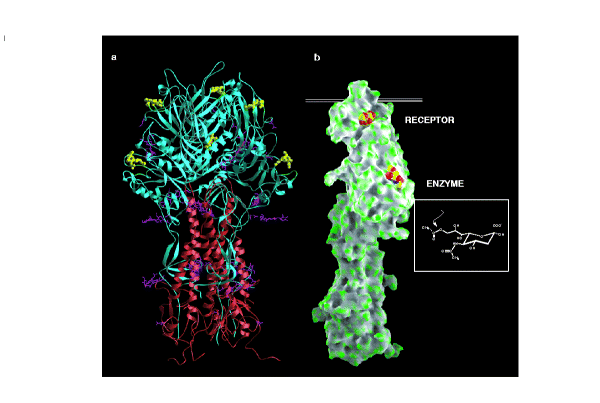
Using cryo-electron microscopy, researchers determined the structures of cross-group broadly neutralizing antibodies from children in complex with different influenza strains. These structural analyses revealed that antibodies from H3-imprinted children exhibited better fit to stalk epitopes on H3 from the imprinting strain relative to H1 from subsequent infecting strains.
The single amino acid at position 46 in the HA2 subunit emerged as crucial: Asp46 in the stalk epitopes of imprinting H3N2 strains and later-infecting H1N1 strains was key to antibody binding, while most pre-pandemic H1N1 strains have Asn46 at this position. This subtle difference—just a single atomic group—promoted a nearly universal shift in the functions of B cell memory targeting the important HA stalk epitope.
Implications for Vaccine Development
These findings have significant implications for influenza vaccine strategies, particularly those targeting the conserved stalk region for universal protection. The research suggests that heterosubtypic imprinting to the HA stalk might be avoided by simultaneous first exposures to H1 and H3 through vaccination in infancy, rather than sequential natural infections.
The study examined infants under one year who received their first dose of influenza vaccine and found they mounted a balanced response to both H1 and H3, with similar cross-reactive responses but without the deleterious reactivity shifts observed after sequential infections. This suggests vaccination timing and strategy could potentially circumvent some imprinting effects.

Future Directions and Considerations
The research opens several important avenues for future investigation. As the authors note, future analyses of larger datasets may resolve rare transcriptional clusters, and evaluation of cell-surface protein markers not evident by transcriptional analysis alone may provide additional insights. The observation that cross-group anti-stalk antibodies from adults were unimpaired suggests that with additional or repeated exposures to H1 strains, H3-imprinted B cells may be outcompeted or adapt through accumulating somatic mutations.
However, the widespread focus on targeting only a few broadly conserved epitopes in influenza vaccine development means that imprinting effects at individual epitope levels are of central concern. As vaccine strategies increasingly target conserved regions like the HA stalk, understanding and potentially mitigating imprinting effects becomes crucial.
Conclusion
The discovery that childhood B cell imprinting specifically impairs antibodies to the haemagglutinin stalk represents a significant advance in our understanding of influenza immunity. This research, detailed in Nature, demonstrates how early life exposures create lasting immune patterns that can either enhance or limit protection against future influenza strains. For vaccine developers, these findings highlight the importance of considering immune history and imprinting effects when designing next-generation influenza vaccines, particularly those targeting conserved regions for broad protection. As we continue to pursue universal influenza vaccines, understanding and addressing immune imprinting will be essential for creating effective, long-lasting protection against this ever-evolving virus.





