Ancient Microbes May Have Used Oxygen 500 Million Years Before the Great Oxidation Event
New research from MIT suggests life on Earth may have evolved the ability to use oxygen hundreds of millions of years before it permanently filled the atmosphere. By tracing the evolutionary history of a key oxygen-processing enzyme, scientists found evidence that aerobic respiration could have emerged during the Mesoarchean era, long before the Great Oxidation Event around 2.3 billion years ago. This discovery challenges traditional timelines and offers a potential explanation for why oxygen accumulation was delayed, indicating early microbes near cyanobacteria may have consumed oxygen as it was produced, slowing its atmospheric rise.
The story of oxygen on Earth is a cornerstone of our planet's history, traditionally marked by the dramatic Great Oxidation Event around 2.3 billion years ago. However, groundbreaking research from the Massachusetts Institute of Technology is rewriting this narrative. A study published in Palaeogeography, Palaeoclimatology, Palaeoecology provides compelling molecular evidence that some forms of life evolved the ability to use oxygen hundreds of millions of years earlier than previously believed. This discovery not only pushes back the timeline for aerobic respiration but also offers a creative solution to a long-standing scientific mystery: if oxygen-producing cyanobacteria appeared around 2.9 billion years ago, why did it take so long for oxygen to build up in the air?

The Great Oxygenation Puzzle
For most of Earth's early history, the atmosphere was devoid of free oxygen. The transformative shift began with cyanobacteria, microbes that developed photosynthesis—the ability to harness sunlight and water, releasing oxygen as a byproduct. Scientific consensus places the emergence of these oxygen producers around 2.9 billion years ago. Yet, geological records show that oxygen did not become a stable, permanent component of the atmosphere until approximately 2.33 billion years ago during the Great Oxidation Event (GOE). This gap of over 500 million years has puzzled scientists for decades. Where did all the early oxygen go? Traditional explanations focused on chemical reactions with rocks and minerals that scrubbed oxygen from the environment. The new MIT study, led by postdoctoral researcher Fatima Husain and associate professor Gregory Fournier, introduces a biological actor into this equation: early life itself may have been consuming the oxygen.
Tracing the Molecular Clock of Respiration
To investigate the possibility of pre-GOE oxygen use, the MIT team focused on a specific biological tool: enzymes. In particular, they studied heme copper oxygen reductases, which are crucial for aerobic respiration as they facilitate the conversion of oxygen into water. This enzyme is found in most oxygen-breathing life today, from bacteria to humans. "We targeted the core of this enzyme for our analyses because that's where the reaction with oxygen is actually taking place," explained Husain. The research strategy involved a sophisticated genetic detective hunt. The team identified the enzyme's genetic sequence and then sifted through massive genomic databases containing millions of modern species to find matching sequences.
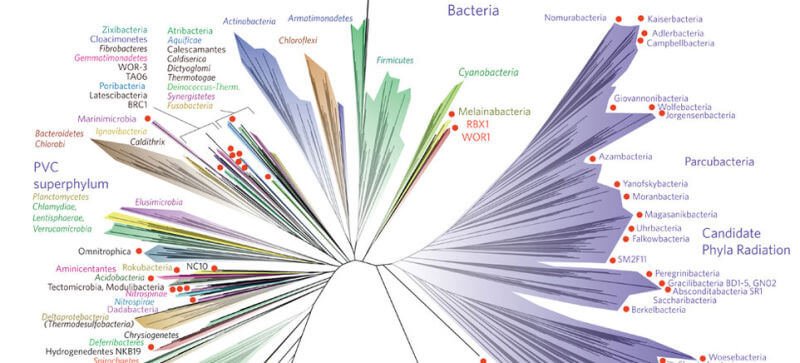
After curating a representative dataset of several thousand species, the researchers placed the enzyme sequences onto an evolutionary tree of life. By using known fossil evidence to anchor specific branches with time estimates, they applied molecular clock techniques to trace the enzyme's origin back through deep time. The computational challenge was significant. "The hardest part of this work was that we had too much data," noted Fournier. "This enzyme is just everywhere... So we had to sample and filter the data down to a dataset that was representative of the diversity of modern life and also small enough to do computation with."
An Archean Origin for Aerobic Life
The results of this painstaking analysis were striking. The molecular clock evidence traced the origin of the heme copper oxygen reductase enzyme back to the Mesoarchean era, which spanned from 3.2 to 2.8 billion years ago. This timeframe is hundreds of millions of years before the Great Oxidation Event. The implication is profound: shortly after cyanobacteria evolved the ability to produce oxygen, other microbial life evolved the sophisticated biochemical machinery to consume it. This suggests that aerobic respiration—a metabolic process considered a later evolutionary innovation—may have its roots in one of Earth's most ancient eras. The microbes capable of this early oxygen use likely lived in close proximity to cyanobacterial mats or blooms, where they could access the small, localized amounts of oxygen being produced.
Reshaping the Narrative of Earth's Oxygenation
This discovery dramatically alters our understanding of the interplay between life and planetary chemistry. It proposes a dynamic where early oxygen production and consumption were closely linked. As cyanobacteria released oxygen, neighboring microbes equipped with the novel enzyme could have immediately used it for energy, effectively acting as a biological sink. This process would have prevented oxygen from leaking out into the broader atmosphere in significant quantities, thereby slowing its global accumulation. "This does dramatically change the story of aerobic respiration," says Husain. "Our study adds to this very recently emerging story that life may have used oxygen much earlier than previously thought. It shows us how incredibly innovative life is at all periods in Earth's history." The research, supported by the Research Corporation for Science Advancement, fits into a broader MIT effort to reconstruct Earth's oxygenation history. By identifying this early biological oxygen sink, the puzzle pieces of the protracted rise of atmospheric oxygen are beginning to fit together more coherently.
The findings underscore a fundamental principle of biogeochemistry: life is not merely a passive passenger on Earth but an active force that shapes its environment. The evolution of oxygen use long before the GOE reveals the metabolic creativity of early microbes and suggests that the transition to an oxygenated world was a more complex, gradual, and biologically mediated process than a single atmospheric 'event.' It highlights how life's adaptations can directly modulate planetary-scale processes, delaying a transformation that would ultimately enable the explosion of complex, multicellular life billions of years later.