How DeepMind's AlphaGenome AI Is Decoding the Mysteries of Rare Diseases
Google DeepMind's AlphaGenome AI is tackling one of biology's greatest challenges: interpreting the 98% of the human genome that doesn't code for proteins. This 'dark matter' of DNA holds crucial clues to rare, often fatal diseases that have eluded diagnosis through conventional methods. By predicting how mutations in non-coding regions affect gene activity, AlphaGenome is providing researchers with powerful new tools to solve medical mysteries that have devastated families worldwide. The technology is already being deployed in diagnostic hackathons where scientists race against time to give answers to those living with undiagnosed conditions.
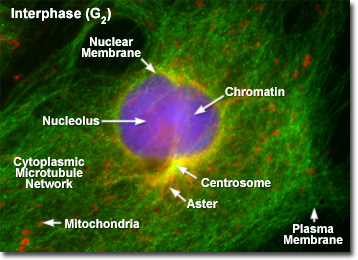
The human genome contains approximately 20,000 protein-coding genes, but these represent only about 2% of our total DNA. For decades, the remaining 98%—often called the 'dark matter' of the genome—has presented one of biology's most formidable challenges. This non-coding DNA, once dismissed as 'junk,' is now recognized as containing crucial regulatory elements that control when, where, and how genes are expressed. When mutations occur in these regions, they can lead to devastating rare diseases that have historically been nearly impossible to diagnose.

Enter AlphaGenome, an artificial intelligence model developed by Google DeepMind that's revolutionizing how researchers approach these diagnostic challenges. As described in a recent Nature publication, AlphaGenome can predict the diverse effects of mutations in non-coding DNA sequences, including how they might affect the activity of nearby genes. This capability represents a significant breakthrough in the quest to understand the genetic basis of rare diseases that affect approximately 350 million people worldwide.
The Diagnostic Challenge of Non-Coding DNA
Traditional approaches to diagnosing rare genetic diseases have focused primarily on mutations in protein-coding regions of the genome, known as the exome. This focus has left a significant diagnostic gap, as many conditions originate from variations in non-coding regions that regulate gene expression. Eric Klee, a bioinformatician at the Mayo Clinic in Rochester, Minnesota, notes that these non-coding variants "often get triaged" in standard diagnostic workflows, leaving patients and their families without answers.
The consequences of this diagnostic gap are profound. Without a diagnosis, patients cannot access targeted treatments, clinical trials, or even basic information about disease progression. Families are left in medical limbo, unable to make informed decisions about care or understand what the future might hold for their loved ones.

AlphaGenome in Action: The Undiagnosed Hackathon
The practical application of AlphaGenome was demonstrated during the Undiagnosed Hackathon held at the Mayo Clinic in September 2025. This event brought together more than 100 researchers who voluntarily "locked themselves in a room" to tackle 29 of the most challenging undiagnosed medical conditions. The hackathon was organized by the Wilhelm Foundation, a Swedish charity founded by Helene and Mikk Cederroth after they lost three of their four children to an undiagnosed disease.
During the three-day event, researchers deployed AlphaGenome to search for genetic causes that had eluded conventional diagnostic methods. The AI model's ability to predict how non-coding variants affect gene expression provided researchers with new hypotheses to test and pathways to explore. As Helene Cederroth emphasizes, "If you don't have a diagnosis, you are left behind." The hackathon represented a concerted effort to ensure that fewer families experience this abandonment.
Validating AlphaGenome's Predictions
Prior to the hackathon, researchers had the opportunity to test AlphaGenome's predictions against known diagnostic cases. Eric Klee and his colleagues used the AI model to analyze a variant they had previously linked to an individual's diagnosis through experimental work. Their laboratory studies had shown that the mutation altered gene expression in cardiac cells but not in neural cells—a finding consistent with the patient's specific symptoms.
Remarkably, AlphaGenome's predictions of the variant's effects supported this conclusion, validating the AI model's accuracy and potential clinical utility. This validation is crucial for building trust in AI-driven diagnostic tools and demonstrates how machine learning can complement traditional laboratory research to accelerate medical discoveries.

The Future of AI in Rare Disease Diagnosis
The development of AlphaGenome represents a significant step forward in the application of artificial intelligence to genomic medicine. By focusing on the non-coding regions of DNA that have traditionally been overlooked, DeepMind's technology opens new avenues for understanding disease mechanisms and developing targeted interventions. As researchers continue to refine these AI models and validate their predictions through experimental work, we can expect more rare diseases to move from the 'undiagnosed' category to the 'understood' category.
The implications extend beyond rare diseases alone. The regulatory principles uncovered through studying non-coding DNA in rare conditions may provide insights into more common diseases and fundamental biological processes. Furthermore, the collaborative model demonstrated by the Undiagnosed Hackathon—bringing together AI developers, bioinformaticians, clinicians, and patient advocates—offers a blueprint for accelerating medical discovery in the genomic era.
As AlphaGenome and similar technologies become more widely available to researchers, the hope is that fewer families will face the uncertainty of an undiagnosed condition. The integration of AI into genomic medicine promises to transform how we approach some of healthcare's most challenging puzzles, bringing us closer to a future where every genetic disease can be identified and understood.