Ancient Microbes May Have Used Oxygen 500 Million Years Before the Great Oxidation Event
New research from MIT suggests life on Earth may have evolved to use oxygen far earlier than previously believed. By tracing a key oxygen-processing enzyme, scientists found evidence that microbes developed aerobic respiration hundreds of millions of years before oxygen permanently filled the atmosphere during the Great Oxidation Event. This discovery challenges traditional timelines and offers a potential explanation for why atmospheric oxygen accumulation was delayed, revealing life's remarkable adaptability even in Earth's earliest chapters.
The story of oxygen on Earth is a cornerstone of our planet's history, traditionally marked by the dramatic Great Oxidation Event around 2.3 billion years ago. However, groundbreaking research from the Massachusetts Institute of Technology is rewriting this narrative. A study published in Palaeogeography, Palaeoclimatology, Palaeoecology presents compelling molecular evidence that some forms of life may have learned to "breathe" oxygen as early as 3.2 to 2.8 billion years ago—hundreds of millions of years before oxygen became a stable component of the atmosphere. This discovery not only pushes back the timeline for aerobic respiration but also provides a fascinating new piece to the puzzle of Earth's early oxygenation.

The Great Oxygenation Puzzle
For decades, scientists have grappled with a significant chronological gap in Earth's history. Fossil and geochemical evidence indicates that cyanobacteria, the first oxygen-producing microbes, evolved around 2.9 billion years ago. These organisms performed photosynthesis, releasing oxygen as a byproduct. Yet, geological records show that oxygen did not begin to permanently accumulate in the atmosphere until approximately 2.33 billion years ago during the Great Oxidation Event (GOE). This leaves a perplexing span of over 500 million years where oxygen was being produced but not building up. The prevailing theory has focused on abiotic processes, suggesting that chemical reactions with iron in rocks and oceans scrubbed the early oxygen from the environment. The MIT study introduces a compelling biological actor into this equation: early life itself may have been consuming the oxygen as fast as it was made.
Tracing the Molecular Clock of Respiration
To investigate the possibility of pre-GOE oxygen use, the research team, led by postdoctoral researcher Fatima Husain and associate professor Gregory Fournier, focused on a specific biological marker: heme copper oxygen reductases. This family of enzymes is fundamental to aerobic respiration, acting as the molecular machinery that allows organisms to convert oxygen into water and harness energy. It is present in nearly all oxygen-breathing life today, from bacteria to humans. The researchers' strategy was to trace the evolutionary origins of this enzyme by analyzing its genetic sequence across the tree of life.
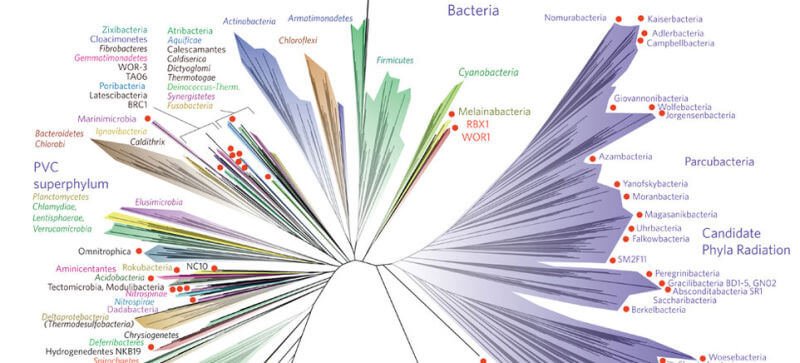
The process involved sifting through massive genomic databases containing millions of species to find matching sequences for the enzyme. After curating a representative dataset of several thousand modern species, the team placed these sequences onto an evolutionary tree. By using known fossil evidence as chronological anchors at various branches, they could apply molecular clock techniques to estimate when the enzyme first diverged and evolved. This meticulous phylogenetic analysis pointed to a clear origin during the Mesoarchean era, between 3.2 and 2.8 billion years ago. This timeline places the advent of this oxygen-utilizing capability squarely in the window after cyanobacteria emerged but long before the GOE.
Implications for Earth's Early History and Life's Ingenuity
The implications of this finding are profound. It suggests that soon after cyanobacteria began their oxygen-producing work, other microbial life in their immediate vicinity evolved the biochemical tools to exploit this new, potent resource. These early aerobic microbes likely lived in localized mats or communities close to cyanobacteria, rapidly consuming the trickle of oxygen as it was produced. This biological consumption would have acted as a global sink, competing with geological processes to keep atmospheric oxygen levels low for an extended period. "This does dramatically change the story of aerobic respiration," says study co-author Fatima Husain. The research paints a picture of an ancient biosphere that was dynamically interactive and highly innovative, with lifeforms quickly adapting to new metabolic opportunities.
This study elegantly connects previously disparate pieces of Earth's history. It provides a plausible biological mechanism for the delayed rise of atmospheric oxygen, complementing existing geochemical models. Furthermore, it underscores a central theme in evolutionary biology: life's exceptional capacity to adapt and diversify in the face of changing environments. The ability to use oxygen, a molecule that was initially toxic to most ancient life, became one of the most successful metabolic strategies on the planet. This research, supported by the Research Corporation for Science Advancement, reminds us that major evolutionary innovations can occur long before their environmental effects become dominant on a planetary scale.
In conclusion, the MIT research offers a transformative view of Earth's oxygenation. It moves the narrative from a simple story of geological and biological succession to a more complex interplay where life actively shaped its own atmospheric environment. The discovery that organisms were using oxygen long before the Great Oxidation Event reveals a deeper layer of resilience and creativity in the history of life on our planet. As Husain notes, these findings help the puzzle pieces fit together, "really underscore[ing] how life was able to diversify and live in this new, oxygenated world" that it helped to create and regulate. The full study can be accessed in the journal Palaeogeography, Palaeoclimatology, Palaeoecology.





